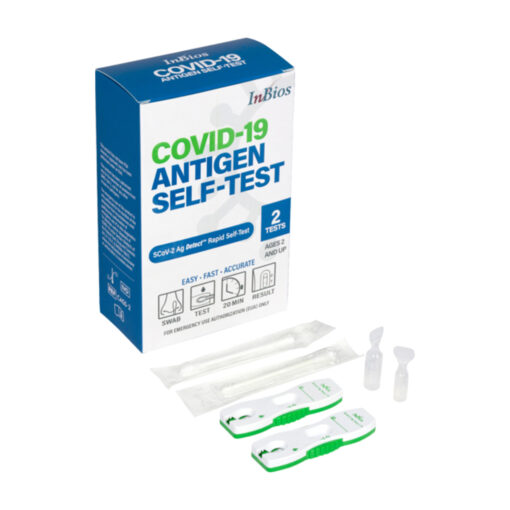
-
- Apparel
- Bariatric
- Dental
- Disinfectants
- Emergency Supplies
- Exam Room
- Eyewear
- Face Masks
- Food & Beverage
- Functional Medicine
- Gloves
- Home Care
- Housekeeping & Janitorial
-
- Promotions | Specials
- Back to School Supplies
- Mens Health
- Urgent Care
- Physician Dispensing
- Compound Pharmacy
- IV Therapy | Infusion
- Weight Loss | Weight Management
- BHRT | Pellet Insertion
- Infection Prevention | PPE | COVID-19
- Aesthetics | Med Spa
- Popular Injectables
- Immunizations | Vaccine Supplies
- At-Home Health Tests
- Cold & Flu Management
- Family Medicine
- Shoebox Shoppers | OCC
- Medical Students
- In Stock Local Anesthetic with EPI
- In Stock Local Anesthetic Plain
- Clinic
- Pediatrics
- OBGYN & Womens Health
- Caregiver Essentials
- Dental
- Hospital